Quality Assurance & Compliance Tracking for Regional Centers
Track, manage, and complete QA reviews with consistency, visibility, and less manual work.

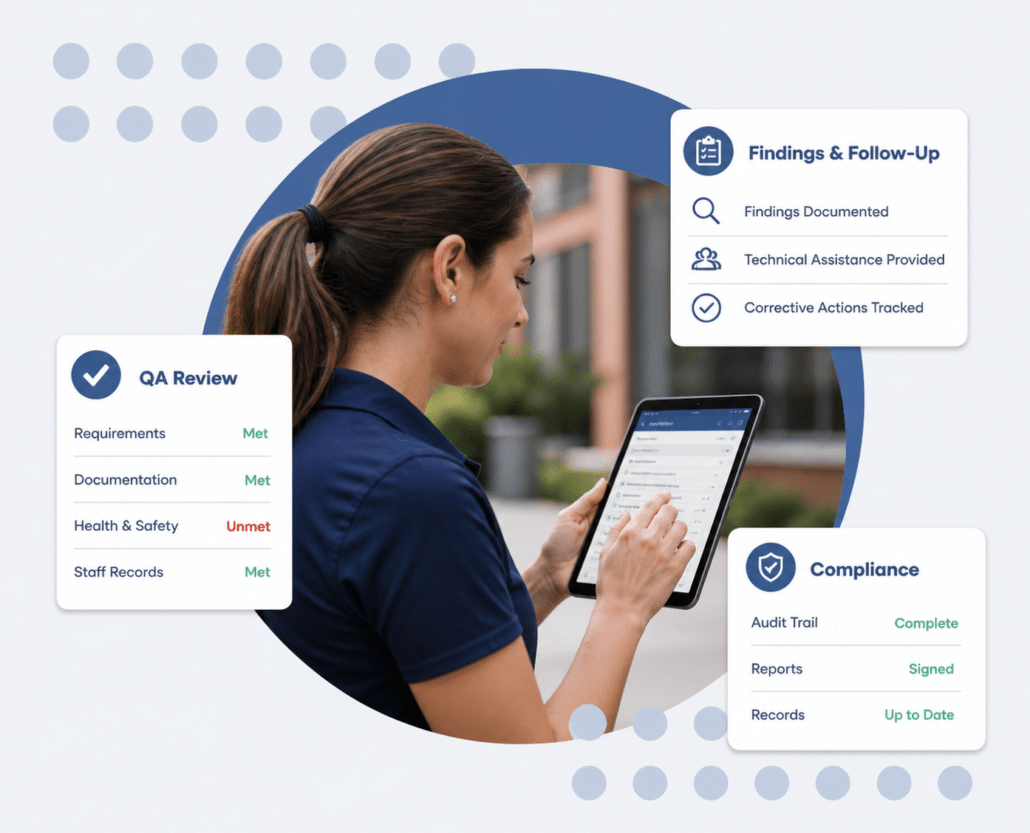
A Better Way to Manage QA & Compliance
Managing QA reviews and compliance often means working across multiple systems, completing reviews manually, and tracking follow-up separately. It works but it’s time-consuming and hard to keep consistent.
QSP brings this work into one place. Teams can complete QA visits in the field, document findings in real time, and track follow-up without recreating or moving information later.
Complete QA Reviews with Structure and Consistency
QA reviews are only as strong as the process behind them. QSP provides structured, customizable forms that guide specialists through each review, ensuring consistency across teams and reducing missed information.
Dynamic Compliance Forms
Create fully customizable QA and compliance forms that reflect your requirements. Forms adjust based on responses (met, unmet, not applicable), prompting the right level of detail and reducing missed information.
Real-Time Data Capture
Capture findings, notes, and required actions during the review, not after. This keeps documentation accurate and eliminates the need to recreate information later.
Field-Ready Mobile Access
Complete QA visits from anywhere using mobile-friendly access. Specialists can enter data, review records, and submit documentation directly from the field.
Configurable Requirements
Define expectations for each requirement, including guidance, references, and outcomes. This helps ensure consistency across reviewers and locations.

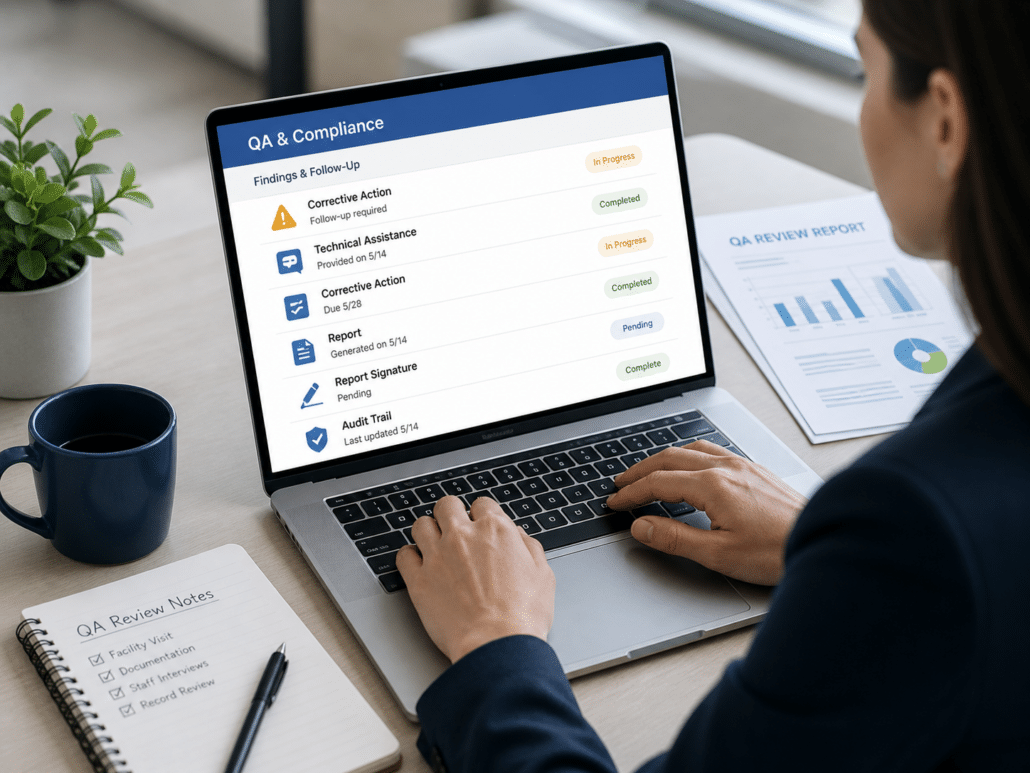
Track Findings, Follow-Up, and Compliance Actions
When issues are identified, follow-up matters. QSP ensures findings, technical assistance, and corrective actions are clearly documented and tracked through completion.
Corrective Action Tracking
Track identified issues through resolution with clear documentation of required actions, timelines, and completion status.
Technical Assistance Documentation
Capture when technical assistance is provided and what support was given, ensuring it’s documented consistently across reviews.
Signable Reports
Generate structured reports that can be reviewed and signed, creating a clear and defensible record of findings and follow-up.
Clear Audit Trails
Maintain a complete, time-stamped record of QA activity, including findings, updates, and follow-up actions for audit readiness.
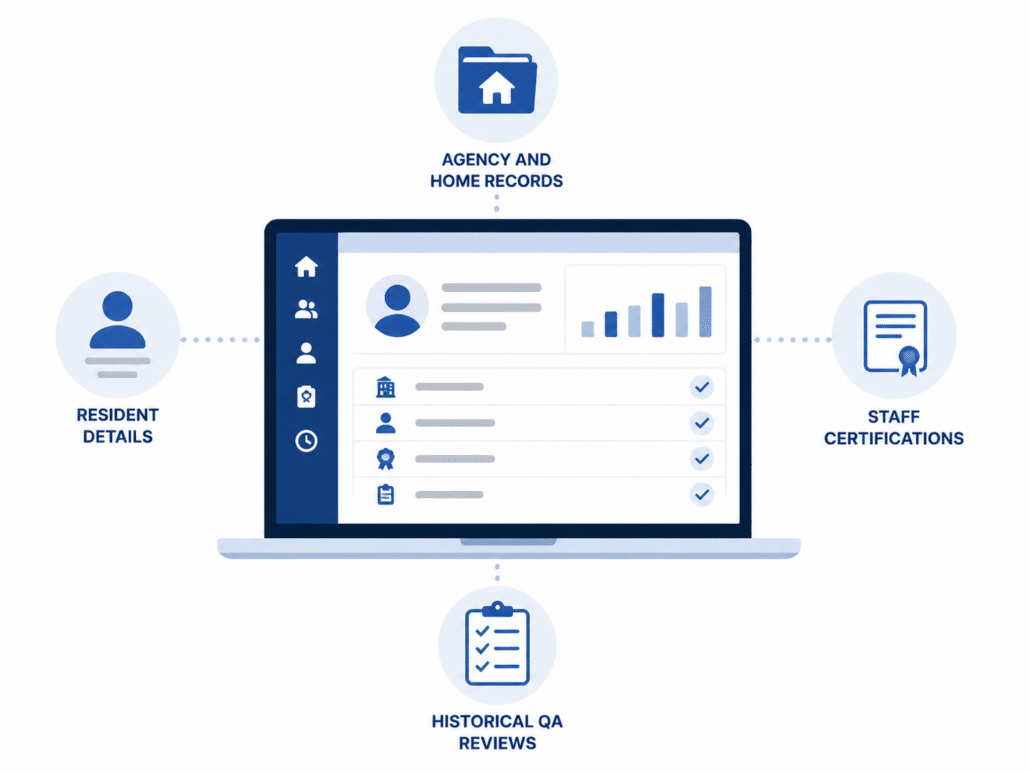
Centralize Provider and QA Data in One Place
Access everything your team needs without switching systems. QSP keeps provider, staff, and QA data organized and accessible, both in the office and in the field.
Agency and Home Records
Store and access key details about agencies and service locations in one centralized system.
Resident Details
Keep relevant resident information available for QA reviews, reducing the need to pull from multiple sources.
Staff Certifications
Track staff records and certifications to ensure required documentation is accessible during reviews.
Historical QA Reviews
View past QA visits and findings to compare changes over time and maintain continuity across reviews.

Standardize Workflows Across Teams
Consistency across QA teams is critical. QSP allows regional centers to define expectations and ensure every review follows the same structure.
Configurable Review Requirements
Set up review criteria that match your standards so every QA visit follows the same structure.
Built-In Guidance and References
Provide reviewers with clear instructions and reference points within each requirement to support consistent decision-making.
Consistent Documentation Across Teams
Ensure that findings, notes, and follow-up actions are captured the same way across all reviewers.
Reduced Variability in Reviews
Minimize differences in how QA is conducted by guiding the process and standardizing expectations.
With QSP, You Can Expect Results Like This

Faster QA Review Completion
Guided forms and mobile access reduce time spent on manual documentation.

More Consistent Documentation
Standardized workflows ensure reviews are complete and aligned across teams.

Better Visibility Across Providers
Centralized data makes it easier to track trends and monitor performance over time.
Frequently Asked Questions
How does QSP support QA and compliance reviews?
QSP provides structured, customizable forms that guide QA specialists through each review. Findings, technical assistance, and corrective actions are documented in real time and tracked through completion.
Can the system be configured to match our regional center’s requirements?
Yes. Forms, workflows, and requirements can be customized to align with each regional center’s QA and compliance standards.
How does QSP improve audit readiness?
All documentation is captured in a structured, time-stamped format with clear audit trails and signable reports, making it easier to demonstrate compliance.
Does QSP work in the field?
Yes. The platform is mobile-friendly, allowing QA specialists to complete reviews and access data during site visits.
Does QSP work in the field?
By centralizing data, standardizing documentation, and guiding the review process, QSP reduces duplicate entry, manual tracking, and follow-up coordination.
